Radiopharmaceuticals provide great promise in the field of cancer diagnostics and treatment. As the demand grows, so supply must ramp up. A critical path for this supply is purification of stable, naturally occurring isotopes of promising elements.
In 2017, Dr Christine Steenkamp’s group at the Physics Department of Stellenbosch University, South Africa, received a Kore TOF-MS from The Council for Scientific and Industrial Research (CSIR) on long-term loan. Kore had manufactured this instrument for CSIR back in 2009 for laser control of molecular dynamics by femtosecond lasers [1,2]. Christine Steenkamp saw the potential to repurpose the instrument for its multiple advantages in the ion detection techniques needed for medical isotope research.
Because different isotopes have different radiopharmaceutical properties, pure samples of the specific isotope are needed. Christine explains, “Different isotopes of the same element have different masses but the same chemical properties, ruling out chemical separation methods. Instead, we rely on methods using laser beams that interact with a vapour and selectively ionize a particular isotope.”
The Kore TOF-MS offered multiple advantages for their experiment. “The low detection limit of the TOF-MS means that we can measure the tiny ion signal while our setup is still far from optimized and use this signal for optimization. The other advantage is that we can measure the full mass spectrum for each burst of laser pulses; therefore, we can see the ratio of the isotopes during optimization in real time.”
Dr Frederick Waso in Christine’s group investigated a laser-based method to purify two isotopes of natural zinc metal (67Zn and 68Zn) that are needed to produce the radiopharmaceuticals used in medical diagnostic PET and SPECT scans [3]; thereby extending earlier experimental work by Dr André de Bruyn [4]. “We work with very small amounts of zinc, evaporating a few milligrams during an experiment”, says Frederick. “We were successful in ionizing the natural isotopes of zinc and demonstrating how the laser pulse energies and pulse timing influenced the ionization efficiency [3,4]. We also developed an ionization scheme that makes it possible to ionize the 67Zn isotope selectively using broadband lasers [3,5]. In our experiment, the abundance of 67Zn was increased from 4% in the natural sample to 90% in the ion sample. The Kore TOF-MS was easy to set up for this method development [6]. The team at Kore is highly knowledgeable about the Kore instruments and gives valuable technical support.”
Once the purification method has been optimized, industry partners can then upscale this for commercial production of high-value medical-grade samples. A medical cyclotron is used to convert the sample into the radiopharmaceutical, which then has a limited lifetime during which it can be used to diagnose or treat patients. This complex, multi-stakeholder chain ends with the patient, who receives state-of-the art medical treatment. Kore is delighted to provide a link in this chain.
Frederick Waso and Christine Steenkamp at Stellenbosch University.
and Christine Steenkamp at Stellenbosch University.
Frederick says, “The Kore TOF-MS was easy to set up for this method development. The team at Kore is highly knowledgeable about the Kore instruments and gives valuable technical support.”
Christine says, “The Kore team has been extremely helpful. When we received the TOF-MS from CSIR in 2017, although this was 8 years since they sold the instrument, the team could provide us with the necessary technical details. They answered questions and provided helpful advice in setting up the instrument.”


Photos of the Kore TOF-MS setup. For laser safety reasons, all optical setups are covered by black-painted panels and laser beams are guided through pipes.

The atomic vapour source, consisting of zinc filled needles heated by electric current.
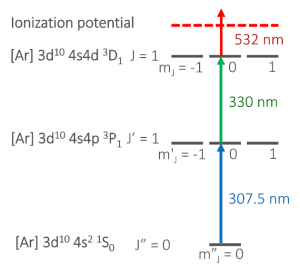
Ionization scheme used for selective ionization of 67Zn. The second excitation step (green arrow) is forbidden for the even-mass isotopes of zinc by quantum mechanical selection rules.
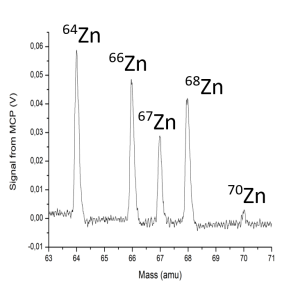
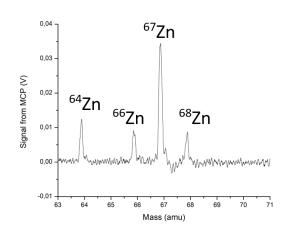
(a) Mass spectrum showing the natural isotopes of zinc. (b) Mass spectrum of zinc isotopes ionised using an ionization scheme where ionization of the even-mass isotopes is suppressed by quantum mechanical selection rules. The ion sample demonstrates an enriched 67Zn when compared to the other isotopes of zinc.
